A Deep Dive into the Heart of Water Treatment
In the realm of water treatment, water softeners play a crucial role in ensuring that the water we use in our homes and industries is free from hardness-causing minerals. At the core of these systems lies a remarkable material known as water softener resin. But how does this resin work? Let's explore the intricate process that makes water softener resin an essential component in water treatment.
Understanding Water Hardness
Before diving into the workings of water softener resin, it's important to understand what water hardness is. Hard water contains high concentrations of calcium and magnesium ions. These minerals can cause a range of issues, from scaling in pipes and appliances to reducing the effectiveness of soap and detergent.
What is Water Softener Resin?
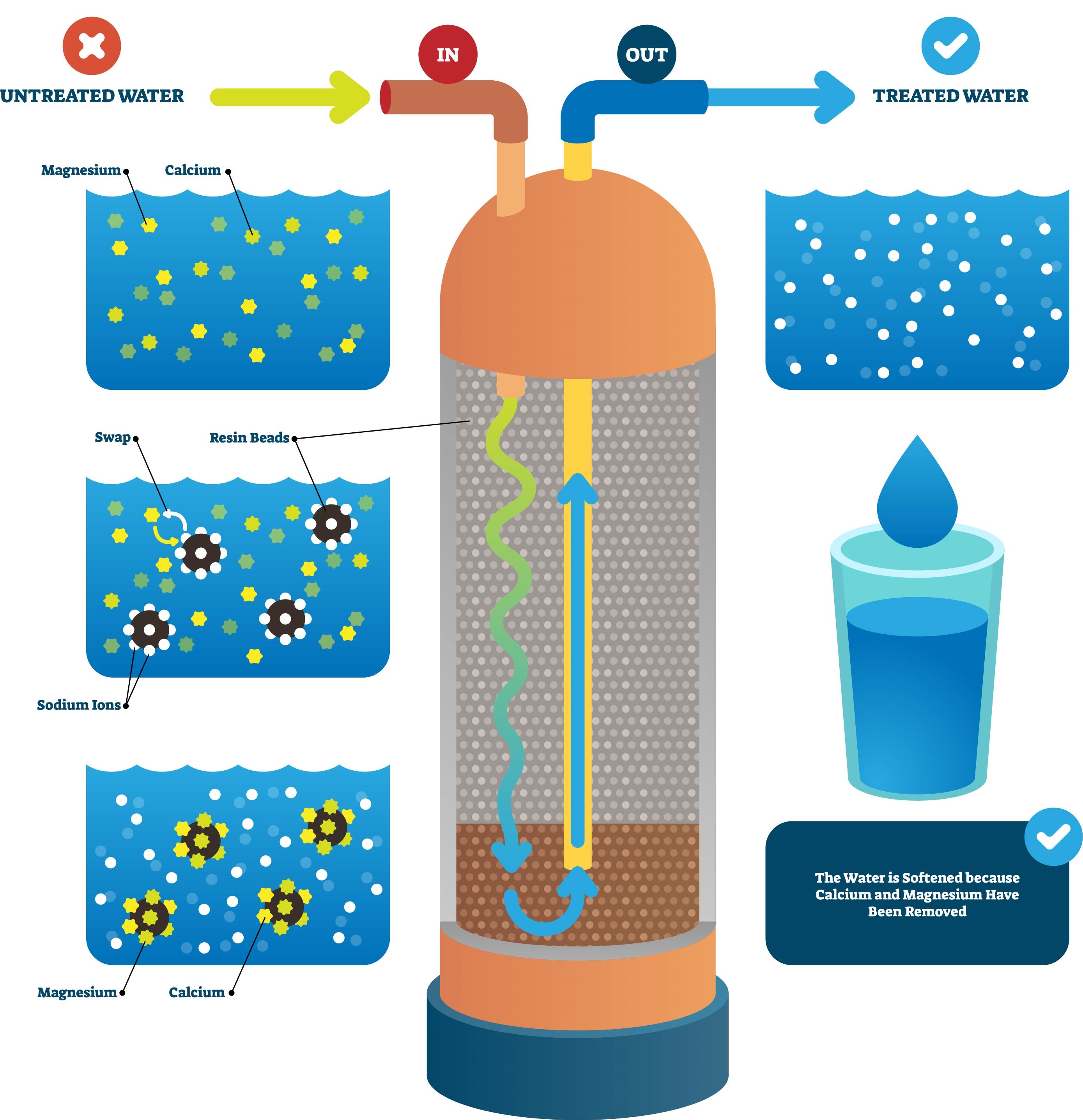
Water softener resin is a synthetic polymer, typically made of polystyrene, that is designed to remove hardness ions from water. These tiny beads, usually about the size of sand grains, are packed into the softener tank. The resin beads are negatively charged and are capable of attracting and holding positively charged ions, such as calcium and magnesium.
The Ion Exchange Process
1. Initiation Phase
When hard water enters the water softener, it passes over the resin beads. The surface of these beads is covered with sodium ions (Na+).
2. Exchange Phase
As the hard water flows through the resin bed, the calcium (Ca2+) and magnesium (Mg2+) ions in the water are attracted to the negatively charged resin beads. The resin beads prefer calcium and magnesium ions over sodium ions, so they exchange their sodium ions for the calcium and magnesium ions in the water.
3. Collection Phase
As a result, the calcium and magnesium ions are trapped on the resin beads, and the sodium ions are released into the water. This process effectively "softens" the water by removing the hardness ions.
4. Regeneration Phase
Over time, the resin beads become saturated with calcium and magnesium ions and lose their effectiveness. To restore their softening capacity, the system undergoes a regeneration process. A brine solution, rich in sodium chloride (salt), is flushed through the resin tank. The high concentration of sodium ions in the brine solution displaces the calcium and magnesium ions from the resin beads, replenishing them with sodium ions.
5. Final Rinse
The displaced calcium and magnesium ions, along with the excess brine solution, are flushed out of the system. The resin beads are now ready to soften water again.
Advantages of Using Water Softener Resin
1. Extended Lifespan of Appliances
By preventing scale buildup, water softener resin helps extend the lifespan of water heaters, dishwashers, washing machines, and other appliances.
2. Improved Soap Efficiency
Softened water enhances the effectiveness of soaps and detergents, leading to better cleaning results and reduced soap usage.
3. Skin and Hair Benefits
Soft water is gentler on skin and hair, reducing dryness and irritation often caused by hard water.
4. Energy Efficiency
Soft water improves the efficiency of heating systems, as there is no scale buildup to impede heat transfer, resulting in energy savings.
Conclusion
Water softener resin is a vital component in the battle against hard water. Through the ion exchange process, it effectively removes calcium and magnesium ions, ensuring that the water we use is soft and free from the issues associated with hardness. Understanding how this remarkable material works can help homeowners and industry professionals appreciate the value of maintaining and properly operating their water softening systems.
By investing in a quality water softener and regularly maintaining the resin bed, you can enjoy the numerous benefits of softened water, from protecting your appliances to enhancing your daily comfort.